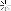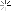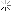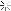Heat treating stuff
I kinda understand how it works. Expose the metal to heat, and it changes naturally, but how does it become this bright and clear?

Is the metal coated in something before heat is added?? Does it have something to do with the type of torch, flame, or temp?


Sorry for the newbie question, ive always been curious. Thanks in advance

Is the metal coated in something before heat is added?? Does it have something to do with the type of torch, flame, or temp?


Sorry for the newbie question, ive always been curious. Thanks in advance

Is it possible to heat treat a material in an argon tank? Is argon flammable? I feel like such a noob 
^^^^p.s.- Oh shiiiit, Kordenbrock huh, I saw your red Hatch in Honda Tuning. Congrats! Nice car!

^^^^p.s.- Oh shiiiit, Kordenbrock huh, I saw your red Hatch in Honda Tuning. Congrats! Nice car!

I worked a for a surgical needle manufacturer, they had a Vacuum heat treat process for stainless and experimented back filling with different gases to add different colors. I dont remember off hand what gases they used. But i remember seeing some needles with a slight red tint and samples that were blue.
Actually we have a belt furance, that used argon as a sheilding gas, and the needles came out blue on the other end. I think the tempertures were in the 1800 range.
Actually we have a belt furance, that used argon as a sheilding gas, and the needles came out blue on the other end. I think the tempertures were in the 1800 range.
Who is Mr Robot?
iTrader: (2)
Joined: Jul 2004
Posts: 21,474
Likes: 10
From: ATL - Where the Pimps and Players dwell
first off, the exhausts pictures are titanium. and i wouldnt call that heat treating. just take a torch and heat to a given temp to give you shiny colors.
Trending Topics
ohh and ask any really good welder all those colors should actually be removed, at least this is what engloid said aka welding god.
As for heat treating a metal what you've shown really isnt heat treating, if you'd like i can go into depth with graphs and tests which I'VE DONE personally to explain it
As for heat treating a metal what you've shown really isnt heat treating, if you'd like i can go into depth with graphs and tests which I'VE DONE personally to explain it

Who is Mr Robot?
iTrader: (2)
Joined: Jul 2004
Posts: 21,474
Likes: 10
From: ATL - Where the Pimps and Players dwell
yea, an ideal weld should be colorless, and if your in the food industy youre not supposed to be able to see any HAZ.
i prefer hardening, but its pointless on an exhuast system, as is heat treating IMO
i prefer hardening, but its pointless on an exhuast system, as is heat treating IMO
<TABLE WIDTH="90%" CELLSPACING=0 CELLPADDING=0 ALIGN=CENTER><TR><TD>Quote, originally posted by Soccerking3000 »</TD></TR><TR><TD CLASS="quote">all those colors should actually be removed</TD></TR></TABLE>
agreed. leaving the color on stainless welds actually reduces the anti-corrosion properties of the base material and weld.
agreed. leaving the color on stainless welds actually reduces the anti-corrosion properties of the base material and weld.
<TABLE WIDTH="90%" CELLSPACING=0 CELLPADDING=0 ALIGN=CENTER><TR><TD>Quote, originally posted by wantboost »</TD></TR><TR><TD CLASS="quote">first off, the exhausts pictures are titanium. and i wouldnt call that heat treating. just take a torch and heat to a given temp to give you shiny colors. </TD></TR></TABLE>
There is your answer
There is your answer
<TABLE WIDTH="90%" CELLSPACING=0 CELLPADDING=0 ALIGN=CENTER><TR><TD>Quote, originally posted by wantboost »</TD></TR><TR><TD CLASS="quote">first off, the exhausts pictures are titanium. and i wouldnt call that heat treating. just take a torch and heat to a given temp to give you shiny colors. </TD></TR></TABLE>
and the last picture, the pipe on the left is after cleaning and passivation/pickling.
and the last picture, the pipe on the left is after cleaning and passivation/pickling.
By the way, heat treating is not to get color, its for strength
http://www.google.com/search?h...title
http://www.google.com/search?h...title
The colors are due to oxides. Welding has very large thermal gradients (liquid metal at fusion plane and progressively cooler as you move away from the weld). Due to the range of temperature the metal experiences the oxide layer thickness and composition varies, causing the rainbow look.
"In steels, there are three common stable oxides: FeO, Fe2O3, and Fe3O4 (sorry no subscripts on the numbers). These three oxides have different formation kinetics and stabilities at different temperatures. " - A.P.
Oxides require oxygen to form. Even with purging and back-purging enough oxygen is still present at the weld location to cause the range of oxides types and thicknesses typically observed.
If the material was exposed to a uniform temperature, the piece would be one solid color. The color of the material can be a crude approximation for determining the termpatures that the metal has seen.
Hope this helps.
~SKY~
"In steels, there are three common stable oxides: FeO, Fe2O3, and Fe3O4 (sorry no subscripts on the numbers). These three oxides have different formation kinetics and stabilities at different temperatures. " - A.P.
Oxides require oxygen to form. Even with purging and back-purging enough oxygen is still present at the weld location to cause the range of oxides types and thicknesses typically observed.
If the material was exposed to a uniform temperature, the piece would be one solid color. The color of the material can be a crude approximation for determining the termpatures that the metal has seen.
Hope this helps.
~SKY~
Thread
Thread Starter
Forum
Replies
Last Post